Effect of Solution Temperature on the Corrosion Behavior of 6061-T6 Aluminum Alloy in NaCl Solution
- PDF / 1,819,948 Bytes
- 8 Pages / 593.972 x 792 pts Page_size
- 66 Downloads / 495 Views
JMEPEG https://doi.org/10.1007/s11665-020-04932-5
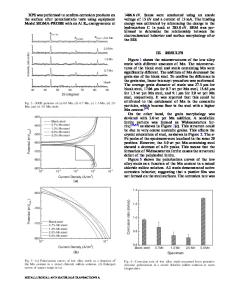
Effect of Solution Temperature on the Corrosion Behavior of 6061-T6 Aluminum Alloy in NaCl Solution M. Zhu, B.Z. Zhao, Y.F. Yuan, S.Y. Guo, and J. Pan (Submitted April 24, 2020; in revised form June 13, 2020) The influence of solution temperature on the corrosion behavior of 6061-T6 aluminum alloy in 3.5% NaCl solution was studied by electrochemical tests, weight loss method, and surface morphology analysis. The results show that the samples treated at different solution temperatures have different corrosion resistance. The anticorrosion resistance of aluminum alloys decreases first and then increases as the solution temperature increases. At 535 °C, the sample has the optimal corrosion resistance, with a slight corrosion degree, while the alloy sample treated at 555 °C shows the most severe degree of corrosion, with a large number of different sized pits and some ulcerous corrosion pits, as well the apparent intergranular corrosion. The corrosion behavior of different solid aluminum alloy samples is closely related to the microstructure. The Mg2Si precipitates act as the anode phase to protect the substrate, and a decrease in the amount of AlFeSi precipitates reduces the number of micro-galvanic corrosion cells, and the severe segregation of AlFeSi precipitates accelerates the corrosion. Keywords
6061 aluminum alloy, corrosion microstructure, precipitated phase, temperature
behavior, solution
1. Introduction 6061 aluminum alloy is a heat treatable strengthening alloy with medium strength and good weldability and machinability in the T6 peak aging state (Ref 1-5). It has been widely used in aerospace, ships, railway vehicles, automobiles, and other fields (Ref 6-9). Under natural conditions, a layer of Al2O3 passive film can be spontaneously formed on the surface of 6061-T6 aluminum alloy to protect the substrate and provide it with a high corrosion resistance. However, different forms of corrosion could be observed during the practical use of aluminum alloys. Scholars have studied how to enhance the corrosion resistance of aluminum alloys using various methods. Shen (Ref 10) found that a micro-arc oxidation coating can improve the corrosion resistance of the 6061 aluminum alloy substrate. Rao (Ref 11) studied the corrosion resistance of aluminum substrate composites with 6061 aluminum alloy as the substrate material and MoS2 as the reinforcement material. The results showed that when 4% molybdenum disulfide (MoS2) was added, it had improved the mechanical properties and corrosion resistance. Since 6061 aluminum alloy ingots and plates are segregated during processing, it is necessary to perform a solution treatment before processing to eliminate the segregation of components in the material and improve the corrosion
M. Zhu, B.Z. Zhao, Y.F. Yuan, and S.Y. Guo, School of Mechanical Engineering and Automation, Zhejiang Sci-Tech University, Hangzhou 310018, China; and J. Pan, National and Local Joint Engineering Research Center of Reliability Analysis and Testing for Me
Data Loading...