Effects of texture and microstructure on the propagation of iodine stress corrosion cracks in zircaloy
- PDF / 3,647,365 Bytes
- 11 Pages / 594 x 774 pts Page_size
- 37 Downloads / 415 Views
I.
INTRODUCTION
Table 1.
F A I L U R E of some fuel elements in light water nuclear reactors has been attributed to stress corrosion cracking (SCC) of the Zircaloy fuel cladding. Mechanical interaction between the fuel pellets and cladding tube generates a tensile hoop stress. Release of volatile fission products, most likely iodine, to the clad ID provides corrosive environment. Experiments to study the effect of crystallographic texture on iodine SCC of Zircaloy have been confined to smooth specimen tests of cladding tubes. Test methods include slotted ring, t differential thermal expansion mandrel, 2 and internal gas pressurization. 3'4 Ambiguity in analyzing a texture effect stems from the inability to separate the crack initiation stage from the crack growth stage and from difficulty in defining the stress and/or stress intensity, K, ahead of a crack propagating through the thin wall fuel cladding. Additional complications arise when one tries to factor out the role of variables such as residual stress, strength differences, heat treatment, manufacturing route, and ID surface condition. The use of conventional fracture mechanics specimens offers several advantages in studying SCC propagation behavior. Complications due to crack initiation are absent. Stress and stress intensity fields ahead of the crack front are well defined. By appropriate choice of plate texture, specimen orientation, and heat treatment, both the texture and microstructure ahead of the propagating crack can be controlled and their effects studied in a systematic manner.
II.
M A T E R I A L S AND TEST PROCEDURES
A. Material Description
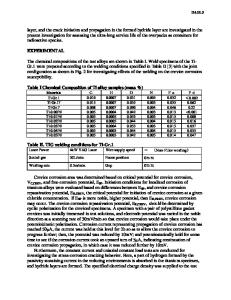
The materials used in the study are plates of Zircaloy-2 and Zircaloy-4 whose ingot compositions are given in Table 1. The fabrication schedules for both plates are shown in
D . B . KNORR, Postdoctoral Associatc, Room 8-326, and R . M . PELLOUX, Professor. Materials Engineering, are both at Massachusetts Institute of Technology, Cambridge, MA 02139. Manuscript submitted December I1, 1980. METALLURGICAL TRANSACTIONS A
Chemical Composition of Zircaloy Plates
Plate Number Type
9908 Zircaloy-2
9912 Zircaloy-4
Sn Fe Cr Ni O
1.51 wt pct 0.14 wt pet 0.10 wt pet 0.05 wt pet 1310 ppm
1.52 wt pet 0.20 wt pet 0. I 1 wt pet 99.8 pct). Any residual water vapor from the iodine column is removed by a molecular sieve (Linde 4A) heated to 155 ---5 ~ to permit passage of the iodine. 12Excess iodine is condensed in a column surrounded by a controlled temperature bath to achieve the desired iodine partial pressure. Only materials known to be inert to iodine attack are used in the test system construction: glass, teflon, and viton Orings. The Ar + 12 gas stream is passed through the glass test chamber containing the preloaded specimens at 0.5 cm 3 per minute. Some attack is evident in the surface of all specimens and reaction product, presumably Zrl4, condenses at the cold ends of the test chamber. Table 4 presents the test matrix for the crack propagation tests. Table 5 gives the test conditions for each specimen.
Data Loading...