In situ transmission electron microscopy and spectroscopy studies of interfaces in Li ion batteries: Challenges and oppo
- PDF / 1,760,503 Bytes
- 7 Pages / 584.957 x 782.986 pts Page_size
- 77 Downloads / 677 Views
W. Xu Energy and Environmental Directorate, Pacific Northwest National Laboratory, Richland, Washington 99352
J. Liu Fundamental and Computational Science Directorate, Pacific Northwest National Laboratory, Richland, Washington 99352
D.W. Choi Energy and Environmental Directorate, Pacific Northwest National Laboratory, Richland, Washington 99352
B. Arey and L.V. Saraf Environmental Molecular Sciences Laboratory, Pacific Northwest National Laboratory, Richland, Washington 99352
J.G. Zhang and Z.G. Yang Energy and Environmental Directorate, Pacific Northwest National Laboratory, Richland, Washington 99352
S. Thevuthasan and D.R. Baer Environmental Molecular Sciences Laboratory, Pacific Northwest National Laboratory, Richland, Washington 99352
N. Salmon Hummingbird Scientific, Lacey, Washington 98516 (Received 15 January 2010; accepted 5 March 2010)
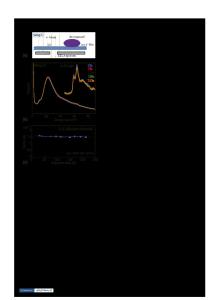
Transmission electron microscopy (TEM) and spectroscopy have been evolved to a stage such that they can be routinely used to probe the structure and composition of the materials with the resolution of a single atomic column. However, a direct in situ TEM observation of structural evolution of the materials in a lithium ion battery during dynamic operation of the battery has never been reported. In this paper, we report the results of exploring the in situ TEM techniques for observation of interfaces in the lithium ion battery during the operation of the battery. A miniature battery was fabricated using a single nanowire and an ionic liquid electrolyte. The structure and composition of the interface across the anode and the electrolyte was studied using TEM imaging, electron diffraction, and electron energy-loss spectroscopy. In addition, we also explored the possibilities of carrying out in situ TEM studies of lithium ion batteries with a solid state electrolyte.
I. INTRODUCTION
Li ion batteries, similar to other electrochemical energy storage devices, are complex multi-component systems that incorporate widely dissimilar phases in physical and electrical contact.1–3 The operation of a lithium ion battery relies critically on electron and ionic a)
Address all correspondence to this author. e-mail: [email protected] DOI: 10.1557/JMR.2010.0198 J. Mater. Res., Vol. 25, No. 8, Aug 2010
http://journals.cambridge.org
Downloaded: 12 Apr 2015
transfer across solid–solid and solid–liquid interfaces and within each of the constituent phases. Repeated charging and discharging of the Li ion battery induces microstructural evolutions both at the interface between the electrolyte and the electrode and within the electrode (active materials) due to Li migration. Although it has been established that this structural evolution of active materials is responsible for the failure of the battery, the mechanisms of the microstructural evolutions as a function of charging/discharging are not well understood.1,2 © 2010 Materials Research Society
1541 IP address: 128.122.253.228
C.M. Wang et al.: In situ transmission electron microscopy and spectroscopy studies of interfaces in
Data Loading...