Influence of aluminum on the martensitic transformation of beta phase CuZn alloys
- PDF / 786,574 Bytes
- 3 Pages / 612 x 792 pts (letter) Page_size
- 95 Downloads / 405 Views
d r i t e s obtainable with s t e r e o s c o p i c viewing is e v e n m o r e s p e c t a c u l a r . This t e c h n i q u e shows p r o m i s e as a tool to study i n c l u s i o n m o r p h o l o g y . 1. D. J. BlJckwede:MetalProgr., 1969, vol.96, pp. 98-102. 2. L. Luyckx:Open Hearth Proceedings TMS-AIME, 1968,vol. 52, pp. 27-34.
200 "4
O
".-
O
o
~ S
_~
~. 10C -
(e/o =t,395) ~ / / - - - C o n s t a n t Zinc
"~
50
(39.5"/., ~'No
Influence of Aluminum on the Martensitic Transformationof Beta PhaseCuZn Alloys HORACE
POPS AND NORMAN
0
A
0.5
I
1.0
"~
1.5
Tronsformolion
l
,
2,0
2.5
Atomic % AI
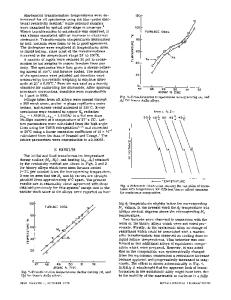
Fig. 1--Martensitie transformation temperatures (M s) vs aluminum content at a constant zinc content (39.5 at. pct) and constant electron concentration (e/a = 1.395).
RIDLEY
T H E m a r t e n s i t i c t r a n s f o r m a t i o n in fl phase CuZn a l loys has been studied in d e t a i l by n u m e r o u s i n v e s t i g a t o r s . 1 In the c a s e of t e r n a r y a l l o y s b a s e d on the b i n a r y fl p h a s e , it has b e e n o b s e r v e d that additions of e l e m e n t s f r o m the s a m e g r o u p of the P e r i o d i c T a b l e have d i f f e r e n t e f f e c t s on the m a r t e n s i t i c t r a n s f o r m a tion t e m p e r a t u r e s , z For e x a m p l e , when the zinc c o n tent is h e l d constant, the t r i v a l e n t e l e m e n t s g a l l i u m and indium l o w e r the M s t e m p e r a t u r e a p p r o x i m a t e l y 90 ~ and 145~ for each at. pct, r e s p e c t i v e l y . B e c a u s e a l u m i n u m is a l s o t r i v a l e n t , it was of i n t e r e s t to c o m p a r e the e f f e c t s of a l u m i n u m additions to a l l o y s with that of g a l l i u m and indium. HORACE POPS is Senior FclIow, Mellon Institute, Carnegie-Mellon University, Pittsburgh, Pa. NORMAN RIDLEY formerly Visiting Fellow, Mellon Institute, is Lecturer, Metallurgy Departmcnt, University of Manchester, Manchester, England. Manuscript submitted March 6, 1970. METALLURGICALTRANSACTIONS
o
Content
o Go
jA,
o AI
25C
9
Go
20(
O
0
15(
E
s
A A
A
~
In
,oc
50
L 0
= J J I 0.5 1.0 1.5 2,0 Atomic % 3rd Element
i 2.5
Fig. 2--Ms temperatures vs third element for CuZn based alloys containing three-valent solutes, e / a = 1.395. VOLUME I, SEPTEMBER 1970-2653
(a)
(c)
(b)
Fig. 3--Thermoelastic and burst-type martensite in a Cu-36.5 Zn-l.50 A1 alloy. Magnification 540 times. The Ms and MB temperatures a r e - 7 0 ~ and -100~ (a) Room Temperature; (b)-83~ (c)-98~ (d)-104~ (e) -118~
(d)
peratures were determined by electrical resistivity m e a s u r e m e n t s and o p t i c a l c o l d s t a g e m i c r o s c o p y . F i g . 1 shows the t e m p e r a t u r e s , M s , c o r r e s p o n d i n g to the s t a r t of the m a r t e n s i t i c t r a n s f o r m a t i o n d u r i n g cooling. F o r a c o n s t a n t z i n c content (39.5 at. pct), the t e m p e r a t u r e is l o w e r e d l i n e a r l y by a p p r o x i m a t e l y 110~ p e r at. p c t of A1. When the e l e c
Data Loading...