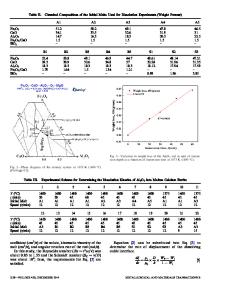
Interface Nanochemistry in Mg-Doped Al 2 O 3 .
- PDF / 402,407 Bytes
- 6 Pages / 612 x 792 pts (letter) Page_size
- 87 Downloads / 432 Views
N2.6.1
Interface Nanochemistry in Mg-Doped Al2O3. Georgette Petot-Ervas1, Gianguido Baldinozzi1, Claude Petot1, Mike J. Graham2, Irwin G. Sproule2 Moussa Tebtoub1 and Jean-Marc Raulot1 1 Laboratoire Structures Propriétés et Modélisation des Solides, CNRS-Ecole Centrale Paris, Grande voie des vignes, F-92295 Châtenay-Malabry cedex, [email protected], [email protected] 2 Institute for Microstructural Sciences NRC, K1A 0R6, Ottawa, Ontario, Canada ABSTRACT In this experimental paper the influence of the grain size, of the thermal history and of the nominal composition on the nanochemistry at the interfaces in α-alumina submicronic powders are analyzed. The consequences on the physical and chemical properties are also discussed. Nanocrystalline Mg-doped α-alumina powders (550 and 1650 ppm by wt MgO) were prepared by the alum process. These powders (10-400 nm) were characterized by X-ray diffraction, SEM, TEM, XPS and surface area measurements (BET). In agreement with BET analysis, SEM observations show that the grain size increases with annealing time and annealing temperature. This effect is less pronounced in the highly doped samples. The less doped samples are also less aggregated and sintering bridges between the grains are still observed. XPS shows that Mg segregates at the periphery of the grains on a depth ≤0.5 nm. The dopant segregation is stronger when the nominal amount of Mg increases, leading eventually to the formation of MgAl2O4 precipitates near the grain surface. It is possible to observe an increase of the dopant segregation with grain size in the low Mg concentration powders. Other impurities like Si or higher cooling rates also favour this Mg segregation. However, the rate of cooling is so effective that it can overshadow all the other parameters (grain size, impurities, ...), suggesting that this is certainly the key parameter for obtaining a perfect sintering of these ceramics, as experimentally shown. INTRODUCTION The fabrication of reliable, high performance ceramics is an important industrial challenge and it has long been recognized that further progress could be made by an increased understanding of the role played by the microstructure and the surface chemistry of the starting powders. Rather surprisingly, little experimental work has been directed towards the powder surface characterization [1]. This may be due to experimental difficulties encountered until recently in characterizing the surface of insulating materials. In previous papers, the authors have already analysed kinetic demixing effects near surfaces in ceramics and have shown the influence of the cooling rate on the near surface cation redistributions [2-6]. Furthermore, XPS confirmed that cation redistributions occur at the periphery of the grains of α-alumina powders doped with magnesium on a depth which depends on the amount of dopant and on the thermal treatment [2]. In this work, a systematic study of cation redistributions near the surface of Mg-doped α-alumina powders is performed to identify the critical parameters
Data Loading...