Origins of Discrepancies Between Kinetic Rate Law Theory and Experiments in the Na 2 O-B 2 O 3 -SiO 2 System
- PDF / 107,685 Bytes
- 10 Pages / 612 x 792 pts (letter) Page_size
- 101 Downloads / 296 Views
Origins of Discrepancies Between Kinetic Rate Law Theory and Experiments in the Na2O-B2O3-SiO2 System B. P. McGrail, J. P. Icenhower, and E. A. Rodriguez Applied Geology and Geochemistry Department, Pacific Northwest National Laboratory, Richland, WA 99352, [email protected] ABSTRACT Discrepancies between classical kinetic rate law theory and experiment were quantitatively assessed and found to correlate with macromolecular amorphous separation in the sodium borosilicate glass system. A quantitative reinterpretation of static corrosion data and new SPFT data shows that a recently advanced protective surface layer theory fails to describe the observed dissolution behavior of simple and complex silicate glasses under carefully controlled experimental conditions. The hypothesis is shown to be self-inconsistent in contrast with a phase separation model that is in quantitative agreement with experiments. INTRODUCTION To predict the long-term fate of waste glass in the subsurface over geologic time scales, a mathematical model that describes glass reactivity is needed. Over the last few decades, a general rate equation has been fashioned to describe the dissolution of glass (and more ordered materials) into aqueous solution [1] v −E r = ka -Hη+ exp a RT where:
Q 1 − Kg
σ
∏aj j
(1)
rv
k aH +
= dissolution rate, g m-2 d-1 = intrinsic rate constant, g m-2 d-1 = hydrogen ion activity
T Q Kg
= temperature, K = ion activity product = pseudoequilibrium constant
aj Ea R
= activity of the jth aqueous species = activation energy, kJ/mol = gas constant, kJ/mol·K
η σ
= pH power law coefficient = Temkin coefficient.
For glasses, where equilibrium with an aqueous solution cannot exist, the value of the pseudoequilibrium constant, Kg, has been assigned to simple silica polymorphs, e.g., amorphous silica or chalcedony [2]. Other investigators have proposed that Kg include other species, such as aluminum, or a complicated activity product of all species found in secondary corrosion products. A final uncertainty rests with the nature and value of the Temkin coefficient, σ, which represents the net reaction order of the decay of the activated complex to the product. Investigators have reported values of σ ranging between 0.4 and 1.0 whereas others have recommended that σ should be ignored in the above equation. We follow the recommendation of Lasaga [3] and assign it a value of 1.0. The rate Equation (1) is the kinetic rate law most often used in performance assessments that treat long-term glass reactivity [4,5]. Recently, a series of papers by French researchers has appeared in the scientific literature that question the validity of Equation (1) for modeling glass dissolution. These researchers have instead advocated a model where an alteration layer on the glass surface is considered a massJJ13.1.1
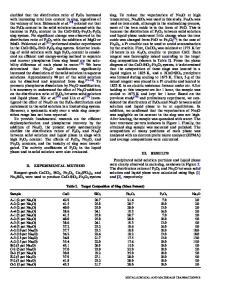
Normalized Release, g/m²
20 transport barrier that ultimately controls the Jégou Glass -1 A/V=400 m , 90°C long-term corrosion rate of silicate glasses. pH(23°C)=9 One important set
Data Loading...