Phase relationships and thermodynamic properties of the Pd-S system
- PDF / 547,622 Bytes
- 6 Pages / 614.28 x 794.28 pts Page_size
- 19 Downloads / 583 Views
I.
INTRODUCTION
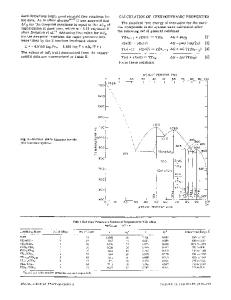
THEinteraction of sulfur with palladium is of importance in a number of industrial applications, for example where Pd is used as a hydrogenation catalyst, and in hydrogen diffusion processes. However, thermodynamic data for the system are very sparse, relying mainly on the work of Niwa et al.,l who measured free energies of formation of Pd4S and PdS by HJH2S equilibration. The dissociation pressures of PdSz and PdS were determined by Biltz and Laar ~and phase diagram measurements were made by Wiebke and Laar, 3 who identified the phases Pd4S, PdS, and PdS2 and a high temperature /3 phase between Pd4S and PdS. A second phase, coming between the/3 phase and PdS, was later found. 4 These two phases were subsequently identified as Pd3S -~ and Pdl6S7,6 and a partially revised phase diagram drawn) In the present work, the phase diagram has been redetermined over the range 0 to 50 at. pct sulfur, and the free energy of formation of Pd4S measured by an emf method. Using this information together with measured sulfur pressures in the liquid matte phase, values for the free energies of formation of Pd3S, Pd16S7, and PdS have been obtained.
II.
EXPERIMENT AND RESULTS
A. Pd-S Phase Diagram Liquidus and other phase transformation temperatures were measured on 25 samples using conventional DTA apparatus, calibrated against the melting points of Au and Ag. The samples, covering a composition range of 13 to 50 at. pct S, and made up from appropriate amounts of PdaS, Pdl6ST, or PdS with Pd metal, were sealed under vacuum into small silica cells. Many samples showed high degrees of supercooling and tended to form metastable phases. Each sample was heated and cooled many times to establish a consistent pattern of behavior, and long annealing times were used to remove metastable phases from the sample where necessary. In particular, samples in the PdI6ST-PdS composition range gave peaks on heating at 829, JEFFREY R TAYLOR is Principal Scientist, Johnson Manhey Research Centre. SonnmgCommon, Reading RG4 9NH, England Manuscript submitted August 8, 1983. METALLURGICALTRANSACTIONSB
898, and 912 K. However, prolonged annealing removed both the 829 and 898 K phase transitions. Samples of composition between Pd3S and Pdt6S7 readily formed Pd3S and PdS on cooling, and an immediate reheating of these samples showed a Pd~S/PdS eutectic at 877 K. However, continued cooling of the samples eventually gave rise to a low temperature phase transition, probably corresponding to the formation of Pd4S. Samples reheated from this point showed only the 829 K transition, and no reaction at 877 K. One anomaly has not been satisfactorily resolved. Both this work and previous work show a peritectic reaction between Pd4S and Pd3S, considered to be at 919 K from this work. This reaction is readily shown by samples containing 20 to 25 at. pct S. However, samples richer in sulfur do not give a reaction at this temperature on heating; they show a well-defined Pd3S liquidus reaction inside the supposed Pd4S/liquid two-phase region--samples app
Data Loading...