Polymorphism in the negative thermal expansion material magnesium hafnium tungstate
- PDF / 302,861 Bytes
- 4 Pages / 585 x 783 pts Page_size
- 57 Downloads / 967 Views
Mark Green National Institute of Standards and Technology, Gaithersburg, Maryland 20899-6102; and Department of Materials Science and Engineering, University of Maryland, College Park, Maryland 20742-2115 (Received 3 August 2007; accepted 25 September 2007)
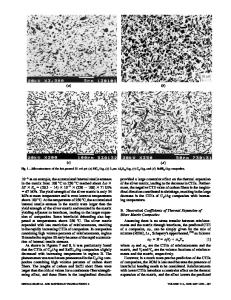
Magnesium hafnium tungstate [MgHf(WO4)3] was synthesized by high-energy ball milling followed by calcination. The material was characterized by variabletemperature neutron and x-ray diffraction. It crystallized in space group P21/a below 400 K and transformed to an orthorhombic structure at higher temperatures. The orthorhombic polymorph adopted space group Pnma, instead of the Pnca structure commonly observed for other A2(MO4)3 materials (A ⳱ trivalent metal, M ⳱ Mo, W). In contrast, the monoclinic polymorphs appeared to be isostructural. Negative thermal expansion was observed in the orthorhombic phase with ␣a ⳱ −5.2 × 10−6 K−1, ␣b ⳱ 4.4 × 10−6 K−1, ␣c ⳱ −2.9 × 10−6 K−1, ␣V ⳱ −3.7 × 10−6 K−1, and ␣l ⳱ −1.2 × 10−6 K−1. The monoclinic to orthorhombic phase transition was accompanied by a smooth change in unit-cell volume, indicative of a second-order phase transition.
I. INTRODUCTION
There has been significant interest in negative thermal expansion (NTE) materials.1–8 Since NTE materials will contract upon heating rather than expand, their incorporation into composites could reduce the overall thermal expansion of the composite. Previous work in the A2(MO4)3 system (A ⳱ trivalent metal, M ⳱ Mo, W) has shown that this thermal expansion behavior is highly dependent on the identity of the A3+ cation.8–11 For trivalent ions up to the size of the smaller rare earths, the A cation is usually six coordinated, and the materials form either monoclinic or orthorhombic structures, the latter being the phase that exhibits NTE. For many compositions, a transition from monoclinic to orthorhombic symmetry is observed upon heating. It has been shown that the A site in A2(WO4)3 compounds can be substituted by a mixture of Hf4+ and Mg2+.12 MgHf(WO4)3 was reported to adopt an orthorhombic structure with space group Pnma at room temperature and 700 °C. Negative thermal expansion along the a and c axes, combined with positive expansion along b, were observed for MgHf(WO4)3, and it was argued that the mechanism of NTE should be the same as for
a)
Address all correspondence to this author. e-mail: [email protected] DOI: 10.1557/JMR.2008.0013 210 J. Mater. Res., Vol. 23, No. 1, Jan 2008 http://journals.cambridge.org Downloaded: 16 Jul 2014
“isostructural” Sc2(WO4)3. However, all previous reports on NTE materials in the A2(MO4)3 family assigned them to space group Pnca. Pnma and Pnca do not share a sub/supergroup relationship; in fact, the only common subgroups of Pnma and Pnca are Pna21 and P21/c. In this paper, we report a detailed structural investigation on MgHf(WO4)3 by powder x-ray and neutron diffraction. The material was found to adopt a monoclinic structure below 400 K, and to transform to an orthorhombic polymorph that is not isostructural to Sc2(WO4)3 at higher temp
Data Loading...