Structural and Luminescence Properties of Pb 2+ -DOPED Li 6 Gd(BO 3 ) 3 Phosphor
- PDF / 747,467 Bytes
- 6 Pages / 594 x 792 pts Page_size
- 95 Downloads / 464 Views
Journal of Applied Spectroscopy, Vol. 87, No. 4, September, 2020 (Russian Original Vol. 87, No. 4, July–August, 2020)
STRUCTURAL AND LUMINESCENCE PROPERTIES OF Pb2+-DOPED Li6Gd(BO3)3 PHOSPHOR E. Erdoğmuşa* and E. Yildizb
UDC 535.37
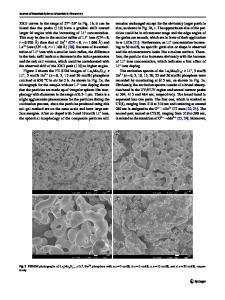
We synthesized pure and Pb2+ ion-doped Li6Gd(BO3)3 phosphors via the solid-state reaction method at 700oC for 5 h in air. The phase analysis of the synthesized materials was determined using DTA/TGA, XRD, and FTIR. The photoluminescence characteristics of pure and Pb2+-doped phosphors were investigated at room temperature using a photoluminescence spectrophotometer. The emission and excitation bands of Pb2+-doped Li6Gd(BO3)3 were observed at 313 and 242 nm, respectively. The dependence of the PL intensities on the Pb2+ concentration for the Li6Gd1–xPbx(BO3)3 (0.005 ≤ x ≤ 0.025) phosphors was also studied. The highest emission intensity was obtained from 0.02 mole Pb2+-doped Li6Gd(BO3)3. Keywords: phosphor, photoluminescence, Li6Gd( BO3)3, Pb2+. Introduction. In recent years, the study of borate crystals and glasses has attracted considerable interest due to their attractive optical and physical properties and wide practical applications. In particular, borate compounds, undoped and doped with rare-earth and transition elements, are promising materials for nonlinear optics, quantum electronics, and laser technology [1–6], scintillators and thermoluminescence dosimeters [7–11], detectors and transformers of ionizing radiation [12, 13], and many other applications [14–16]. From the technological point of view, glassy borate compounds are more promising materials than their crystalline analogues. The growth technology of the borate single crystals is a difficult, longterm, and very expensive process, whereas large high-quality samples of borate glasses can be obtained in a simple, fast, and inexpensive way [17]. The Pb2+ ion, as a well-known dopant for many different matrices, is of great scientific and technological interest. Due to the diversity of its luminescent properties, it provides the possibilities of fabricating novel luminescent materials. Usually, the luminescence of the Pb2+ ion (6s2) is characterized by the ground state 1S0 and two excited states of singlet 1 P1 and triplet 3P0,1,2 transitions, which originate from the 6s2–6s16p1 interconfigurational transition [18–20]. It has been shown that the effect of the s2 ions on the ground state considerably complicates the situation (e.g., Jahn–Teller effect) [18]. Furthermore, the interaction of Pb2+ ions with the host lattice strongly influences its luminescent properties [21, 22]. Double orthoborate Li6RE(BO3)3 (RE = trivalent rare earth ions) crystals are attractive matrix materials owing to their high transparency in the UV and high damage threshold upon exposure to energetic radiations [23]. When activated with RE ions, these materials can also be used as scintillation detectors for thermal neutrons and gamma photons. Li6RE(BO3)3 compounds are known to crystallize in the monoclinic system with space group P21/c and have an is
Data Loading...