The kinetics of the exchange of cobalt(II) ions between NaCl and Na 2 O-SiO 2 -Fe 2 O 3 melts in the temperature range 1
- PDF / 511,766 Bytes
- 7 Pages / 603.28 x 783.28 pts Page_size
- 63 Downloads / 372 Views
NaC1-C~ Na20_SiO2_Fe203
NaC1-CoC12
Co(+)
Distribution coefficients a of the Co 2§ ions between the salt phase and silicate melt are of the 10-2 order. Differential equations describing the transfer process of the Co 2§ ions from the salt phase to the silicate phase were solved. The diffusion coefficients Dco2. in the Na20-SiO2-Fe203 melt were determined by substituting the Co 2§ ion concentration change in fused NaC1 into the differential equations. The values of Dco2. are about 10 -5 cm2s - ~and they decrease with decreasing temperature and increasing Fe203 content in slags. The diffusion coefficient of the cobalt (II) ions in slags determined in this way is consistent with that determined by the rotating disk method26
FUSEDsalts m a y be applied for the extraction of certain metals from liquid slags/,2Studies have shown that amounts of copper, cobalt and other metals m a y be recovered from liquid slags by extraction with fused sodium chloride. TM The present paper deals with the equilibrium and kinetics of the cobalt (II) ion transfer from fused sodium chloride to the slag. As a model synthetic slag some low-melting melts from the N%OSiO2-Fe203 were used whose physicochemical properties have been summarized previously? ,6 Studies were carried out with three types of slags whose compositions are provided in Table I. The system consisting of dilute cobalt (II) chloride in fused sodium chloride and liquid slag from the Na20-SiO2-Fe203 system consists of two practically immiscible phases. In order to determine the distribution coefficients of the cobalt ions and transfer kinetics of these ions from the salt phase to the slag phase, the emf of the following galvanic cell system was measured: (-)Co
NaC1-C~ (x') NaEO-SiO2-Fe203
NaC1-CoC12 (xo)
Co(+) [1]
EXPERIMENTAL The slags were prepared by melting suitable SiO2, Fe203 and Na2CO 3 mixtures (pa). The melts were kept at about 1400 K to remove all carbon dioxide. Sodium chloride (pa) and anhydrous cobalt (II) chloride (pa) J. GOSPOg is Research Associate and A. BOGACZ is Associate Professor in Institute of Inorganic Chemistry and Metallurgy of Rare Elements, and J. SZUST is Research Associate in Institute of Chemical Engineering and Heating Equipment, all with Technical University of Wroclaw,Wybrze~eWyspiafiskiego27, 50-370 Wroctaw, Poland. Manuscript submitted January 14, 1980.
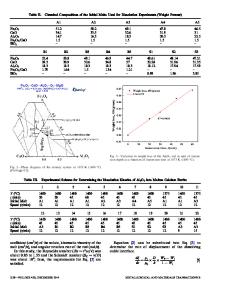
were fully dehydrated by melting in a stream of dry hydrogen chloride. The cell for emf measurements of the concentration cell is shown in Fig. 1. A corundum crucible containing synthetic slag and NaC1 + CoC12 melt was placed in a resistance furnace. The crucible was closed with a glass head. The cobalt electrode immersed in the salt melt was the negative electrode. The crucible was provided with a thickwalled tube of high-alumina material which, together with the salt melt and cobalt electrode functioned as a positive half-cell. The alumina tube was provided with a capillary hole which was plugged with asbestos. When immersed in the melt this became saturated with salt forming a salt br
Data Loading...