Thermodynamics and Kinetics of Bulk Metallic Glass
- PDF / 354,702 Bytes
- 4 Pages / 585 x 783 pts Page_size
- 46 Downloads / 444 Views
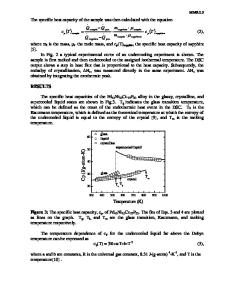
contributions to glass-forming ability (GFA), it is found that BMGs show both unusually high viscosities and low driving forces for crystallization.
Thermodynamics of Supercooled BMG Liquids
and Kinetics of Bulk Metallic Glass
R. Busch, J. Schroers, and W.H. Wang Abstract Bulk metallic glasses (BMGs) are multicomponent alloys with typically three to five components with large atomic size mismatch and a composition close to a deep eutectic. Packing in BMG liquids is very dense, with a low content of free volume resulting in viscosities that are several orders of magnitude higher than in pure metal melts. The dense packing accomplished by structural and chemical atomic ordering also brings the BMG-forming liquid energetically and entropically closer to its corresponding crystalline state. These factors lead to slow crystallization kinetics and consequentially to high glass-forming ability. This article highlights the thermodynamic and kinetic properties of BMGs and their contributions to extraordinarily high glass-forming ability. Some possible links with mechanical properties are also suggested.
Introduction
620
supercooling. The time at the nose is about 60 s, which is amazingly long, even if compared with phase transformations within the solid state such as the austenite–pearlite transformation in steels. By separating the kinetic and thermodynamic
1100 Temperature (K)
Liquids that can yield bulk metallic glasses (BMGs) are multicomponent eutectics or close-to-eutectic alloys with large negative heats of mixing. BMG-forming liquids thus have very low melting points compared with each constituent element, indicating that the melt is thermodynamically quite stable and highly favored with respect to the competing crystalline phases. Even in the supercooled (undercooled) liquid state, they show high thermal stability (i.e., resistance to crystallization). For some BMGs, this stability enables measurements of specific heat,1,2 diffusion,3–5 viscosity,6–8 local atomic motion,9 and emissivity10 in a temperature region that was previously not accessible. The stability of the supercooled liquid also enables experimental determination of the crystallization process,11 which was found to change from a nucleation-controlled mechanism at high temperatures (low undercoolings) to a growth-controlled process at low temperatures.12 Figure 1 shows the experimentally determined time–temperature–transformation (TTT) diagram for crystallization of the supercooled liquid for Zr41.2Ti13.8Cu12.5Ni10.0Be22.5 (Vitreloy 1, or V1).11,13 It shows the typical “nose” shape, which is the result of increasing driving force for crystallization and decreasing atomic mobility upon
Tliq
1000
Zr41.2Ti13.6Cu12.5Ni10.0Be22.5
900 Crystalline 800
Supercooled liquid
700 600
Tg
Glass 1
2 3 Log Time (s)
Figure 1. Time–temperature– transformation diagram for the primary crystallization of Vitreloy 1. Data obtained by electrostatic levitation (●)11 and processing in high-purity carbon crucibles (●)13 are included. Calculated times for a cryst
Data Loading...