V 2 O 5 -P 2 O 5 -Fe 2 O 3 -Li 2 O Glass-Ceramics as High-Capacity Cathode for Lithium-Ion Batteries
- PDF / 2,811,007 Bytes
- 5 Pages / 612 x 792 pts (letter) Page_size
- 42 Downloads / 470 Views
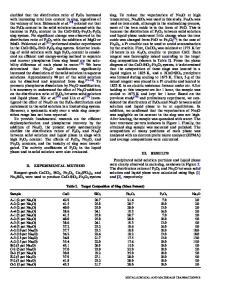
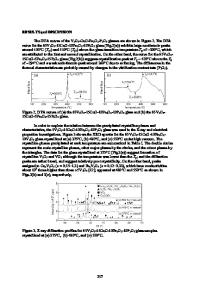
V2O5-P2O5-Fe2O3-Li2O Glass-Ceramics as High-Capacity Cathode for Lithium-Ion Batteries Takuya Aoyagi1, Tadashi Fujieda1, Kazutaka Mitsuishi2, Jun Kawaji1, Tatsuya Toyama1, Kazushige Kono1 and Takashi Naito1 1 Hitachi Research Laboratory, Hitachi, Ltd., 7-1-1 Omika-cho, Hitachi-shi, Ibaraki-ken 3191292, Japan 2 National Institute for Materials Science, 1-2-1 Sengen, Tsukuba, Ibaraki 305-0047, Japan ABSTRACT We discuss the advantages of V2O5-P2O5-Fe2O3-Li2O glass-ceramics as a cathode for lithium-ion batteries. The glass was prepared by using the melt quenching method. The glassceramics were produced by heat treatment in air. LixV2O5 crystal was only confirmed as the precipitated phase and the degree of crystallinity was approximately 90%. The total capacity of the glass-ceramics was 340 Ah/kg at a C/20 rate for 1.5-4.2 V cutoff ranges. It is 10% higher than the capacity of the glass cathode. Moreover, the charge-discharge performance of the glassceramics cathode showed good cycleability similar to that of the glass. The glass-ceramics had a 83% capacity retention after 40 cycles. These results show that glass-ceramics is a potential candidate for lithium-ion cathode materials. INTRODUCTION Vanadium based materials such as V2O5, LiV3O8, and LixV2O5 has been extensively studied as a cathode for Li-ion batteries [1-3]. Their open structure combined with wide range of oxidation states for vanadium, which range from V5+ to V2+, allows for a high theoretical specific capacity. The theoretical capacity of LixV2O5 is 442 Ah/kg when x=3 [3-5]. It is about three times higher than that of LiCoO2. However, LixV2O5 has an irreversible structural change when Li insertion is more than x=1 [6,7]. Several studies on how to improve this structural reversibility have been done [8-11]. Vitrification is a way of improving electrochemical performance. Several studies showed that glass materials such as V2O5-P2O5 [12], TeO2-V2O5 [13], Li2O-V2O5-P2O5 [14] exhibited good rechargability. In particular, the V2O5-P2O5 glass system has a high capacity. However, this system has poor water durability [15] and the capacity of it seems a little lower than that of V2O5 crystals. In this work, we discuss the advantages of glass ceramics over glass materials. We show how using glass-ceramics improves electrochemical performance.
EXPERIMENT Glass was prepared by using the melt quenching method, and V2O5, P2O5, Fe2O3 and Li2CO3 powders were used for the glass synthesis. The mixed powders were melted at1273 K in a platinum crucible and kept at the temperature for one hour. The molten glass was cast onto a stainless steel plate at 423 K. The resulting materials were fully amorphous as was confirmed by conducting an X-ray diffraction (XRD) analysis using a diffractometer system equipped with monochromatic CuKα (Rigaku, model RINT-2000). The crystallization tendency was measured with a differential thermal analysis (DTA) at a heating rate of 5 K/min using milled glass powders. The prepared glasses were crystallized by an electric furnace for 2 hours (the h
Data Loading...