Vapor pressure and thermodynamic study of the Co-Ni system
- PDF / 493,053 Bytes
- 6 Pages / 612 x 792 pts (letter) Page_size
- 24 Downloads / 407 Views
IN
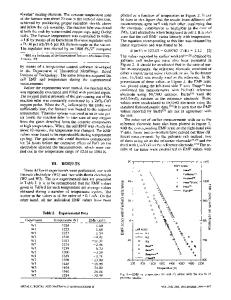
o u r p r e v i o u s p a p e r1 we h a v e r e p o r t e d t h e r m o d y n a m i c f u n c t i o n s i n t h e C o - N i s y s t e m by t h e i s o t o p e exchange method. T h e partial thermodynamic funct i o n s for cobalt were m e a s u r e d directly, w h e r e a s t h e v a l u e s f o r n i c k e l w e r e c a l c u l a t e d by i n t e g r a t i n g t h e G i b b s - D u h e m e q u a t i o n . T h e r e s u l t s s h o w e d that n e g a t i v e d e v i a t i o n s f r o m R a o u l t ' s l a w p r e v a i l in t h e C o - N i s y s t e m . A n a t t e m p t was made to i n t e r p r e t t h e s e r e s u i t s in t e r m s o f o r d e r i n g p r o c e s s e s in t h e C o - N i syst e m u s i n g t h e B i r c h e n a l l m o d e l ,e I n t h i s s y s t e m s h o r t r a n g e o r d e r c a n be a s s u m e d a c c o r d i n g t o t h e P u z e y a n d G o m a n ' k o v m e a s u r e m e n t s3 o f t h e m a g n e t i c a n i s o tropy constant. On the o t h e r h a n d , however, neutron d i f f r a c t i o n 3 did not i n d i c a t e t h e e x i s t e n c e of a l o n g o r s h o r t - r a n g e o r d e r , a n d t h u s the q u e s t i o n o f o r d e r i n g p r o c e s s e s n e a r the N i a C o c o m p o s i t i o n r e m a i n s o p e n . W i t h r e g a r d t o the c o m p l i c a t i o n s c o n n e c t e d w i t h the i s o t o p e e x c h a n g e m e t h o d ,4 the t e m p e r a t u r e d e p e n d e n c e o f t h e cobalt v a p o r p r e s s u r e was d e t e r m i n e d f r o m a s m a l l n u m b e r of p o i n t s o n l y a n d t h e r e f o r e the a c c u r a c y of the v a p o r p r e s s u r e v a l u e s e v a l u a t e d i n the p r e v i o u s p a p e r w a s not too h i g h ( f r o m 30 t o 55 p c t - - s e e R e f . 1 , T a b l e II). In t h i s w o r k , we h a v e p e r f o r m e d a n e w s e r i e s of m e a s u r e m e n t s o f c o b a l t v a p o r p r e s s u r e by the e f f u s i o n m e t h o d to a c h i e v e m o r e a c c u r a t e v a l u e s a n d to v e r i f y t h e r e p r o d u c i b i l i t y of t h e r e s u l t s o b t a i n e d by b o t h m e t h o d s . F u r t h e r , we h a v e c a r r i e d o u t a n o t h e r s e r i e s of m e a s u r e m e n t s f o r t h e s e c o n d c o m p o n e n t ( n i c k e l ) o f the N i - C o s y s t e m by t h e e f f u s i o n m e t h o d i n o r d e r t o a v o i d the i n a c c u r a c i e s c a u s e d by i n t e g r a t i n g the G i b b s - D u h e m e q u a t i o n .
EXPERIMENTAL T h e evaporation rate w a s m e a s u r e d for pure cobalt, pure nickel and the C o - N i alloys w h i c h were kindly provided by the C e n t r e National de R e c h e r c h e s M d t a l lurgiques Liege, B e l g i u m . The compositions of the alloys are given in T a b l e I. T h e cobalt used in t h e s e alloys contained the following impurities: 0.016 wt pct Fe, 0.014 wt pct Cu, 0.01 wt pct M n . O t h e r e l e m e n t s w e r e present in m u c h l o w e r concentrations. Nickel used for the preparation of s a m p l e No.
Data Loading...