A thermodynamic analysis of the Fe-Mn-C system
- PDF / 884,407 Bytes
- 9 Pages / 612 x 792 pts (letter) Page_size
- 54 Downloads / 437 Views
THE
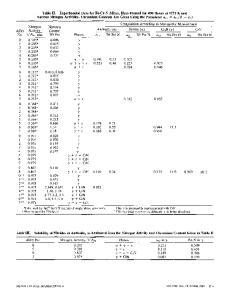
p h a s e equilibria in t h e F e - M n - C s y s t e m h a v e b e e n s t u d i e d by m a n y i n v e s t i g a t o r s . V o g e l a n d D S r ing1 f i r s t published a s t u d y covering t h e w h o l e r a n g e of composition from Fe-C t o M n - C . More detailed s t u d i e s w e r e l a t e r c a r r i e d o u t by I s o b e2 a n d K u o a n d P e r s s o n3 a n d t h e m a i n f e a t u r e s of t h e p h a s e d i a g r a m were thus established. D u r i n g t h e last d e c a d e t h e r e has been a n i n c r e a s i n g i n t e r e s t in t h e t h e r m o d y n a m i c p r o p e r t i e s a n d in the relation between them and the p h a s e d i a g r a m . T h i s l i n e o f r e s e a r c h w a s s t a r t e d by N i s h i z a w a4 w h o s t u d i e d t h e c a r b o n a c t i v i t y of a u s t e n i t e c o n t a i n i n g m a n g a n e s e a n d t h e d i s t r i b u t i o n of m a n g a n e s e b e t w e e n a u s t e n i t e a n d c e m e n t i t e (M3C) o r M23C6 a t 1273 K . H e d e d u c e d t h e f r e e e n e r g y o f t h e m i x e d c a r b i d e s ( F e , Mn)3C a n d ( F e , Mn)2sC6 a n d c a l c u l a t e d t h e p h a s e d i a g r a m . C o n t r a r y to e a r l i e r i n v e s t i g a t o r s , N i s h i z a w a c o n c l u d e d that t h e a u s t e n i t e e x t e n d s a l l t h e w a y f r o m t h e F e - C side t o t h e M n - C side of t h e t e r n ary d i a g r a m . Nishizawa's experimental d a t a w e r e l a t e r r e i n t e r p r e t e d by N i s h i z a w a a n d U h r e n i u s 5 u s i n g a n e w t h e r m o d y n a m i c m o d e l f o r t h e a u s t e n i t e but t h e m a i n f e a t u r e s r e m a i n e d u n c h a n g e d . A n e w s t u d y of t h e c a r b o n a c t i v i t y o f F e - M n - C a u s t e n i t e was made by W a d a e t a l~ w h o e x t e n d e d t h e t e m p e r a t u r e r a n g e . T h e i r r e s u l t s a r e in c l o s e a g r e e m e n t w i t h N i s h i zawa's. A n e w e x t e n s i v e s t u d y o f t h e F e - M n - C s y s t e m was r e c e n t l y m a d e by B e n z e t a l .7 T h e y i n v e s t i g a t e d t h e p h a s e d i a g r a m b e t w e e n 873 a n d 1373 K a n d , i n p a r t i c u l a r , t h e y e s t a b l i s h e d t h e s o l u b i l i t y o f c a r b o n i n aust e n i t e . I n agreement with Nishizawa t h e y concluded that austenite can extend from Fe-C t o Mn-C and t h e i r data for the distribution of M n b e t w e e n austenite a n d M~C o r M23C6 w e r e i n e s s e n t i a l a g r e e m e n t w i t h Nishizawa's. However, their experimental solubility o f c a r b o n in a u s t e n i t e was s i g n i f i c a n t l y l o w e r t h a n t h e c a l c u l a t e d s o l u b i l i t y a c c o r d i n g t o N i s h i z a w a4 a n d Nishizawa a n d Uhrenius.5 Benz et a l made a n e w t h e r m o d y n a m i c e v a l u a t i o n , u s i n g t h e i r own d a t a o n t h e c a r b o n a c t i v i t y , t h e s o l u b i l i t y o f c a r b o n a n d t h e dist r i b u t i o n of M n . H o w e v e r , t h
Data Loading...