A thermodynamic investigation of the Ag-Mn system
- PDF / 1,166,134 Bytes
- 12 Pages / 612 x 792 pts (letter) Page_size
- 57 Downloads / 500 Views
POOL
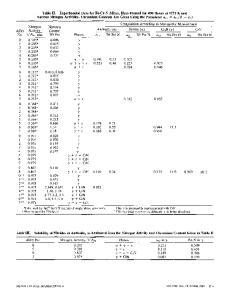
A t h e r m o d y n a m i c study has b e e n made of the A g - M n s y s t e m in the t e m p e r a t u r e r a n g e of 1100 ~ to 1200~ u s i n g a modified K n u d s e n effusion t e c h n i q u e . The a c t i v i t i e s of s i l v e r and m a n g a n e s e w e r e d e t e r m i n e d by a n a l y s i s of the v a p o r phase u s i n g X - r a y f l u o r e s c e n c e and a p p r o p r i a t e g r a p h i c a l i n t e g r a t i o n of the c o m p o s i t i o n function r e l a t i n g a c t i v i t y and v a p o r phase c o m p o s i t i o n . S i l v e r - r i c h s o l u t i o n s , in the s i n g l e - p h a s e r e g i o n , w e r e found to exhibit e x o t h e r m i c heats of m i x i n g and n e g a t i v e d e v i a t i o n s f r o m i d e a l i t y for the a c t i v i t y of the s o l v e n t and both n e g a t i v e and p o s i t i v e d e v i a t i o n s f r o m R a o u l t ' s law for the a c t i v i t y of the solute. M a n g a n e s e - r i c h s o l u t i o n s , in the t w o - p h a s e r e g i o n , exhibit e n d o t h e r m i c heats of m i x i n g and p o s i t i v e d e v i a t i o n s f r o m i d e a l i t y for the a c t i v i t i e s of both the solute and s o l vent. The n e g a t i v e e x c e s s e n t r o p i e s of m i x i n g in the dilute m a n g a n e s e r e g i o n a r e a t t r i b uted to m a g n e t i c and e l e c t r o n i c c h a n g e s which o c c u r at 20 at. pct Mn. T h e s e changes in m a g n e t i c and e l e c t r o n i c p r o p e r t i e s a r e a r e s u l t of a g r a d u a l change in the M n - M n i n t e r a t o m i c s e p a r a t i o n and e l e c t r o n i c s t r u c t u r e . With i n c r e a s i n g m a n g a n e s e c o n t e n t beyond 20 at. pct Mn, the e x c e s s e n t r o p i e s b e c o m e p o s i t i v e , p a s s i n g t h r o u g h a m a x i m u m a s the t w o - p h a s e b o u n d a r y is r e a c h e d . The p o s i t i v e e x c e s s e n t r o p i e s of m i x i n g a r e a t t r i b u t e d to lattice d i s t o r t i o n r e s u l t i n g f r o m s i z e d i f f e r e n c e s between s i l v e r and m a n g a n e s e a t o m s .
A g e n e r a l u n d e r s t a n d i n g of the t h e r m o d y n a m i c s t a b i l i t y of m e t a l s and a l l o y s has come to be r e c o g n i z e d as an i m p o r t a n t tool for m e t a l l u r g i s t s both f r o m the standpoint of i n d u s t r i a l a p p l i c a t i o n s and t h e o r e t i c a l i n t e r e s t . The d e v e l o p m e n t of g e n e r a l i z e d s o l u t i o n t h e o r i e s in r e c e n t y e a r s has led to a b a s i c u n d e r s t a n d ing of a l l o y b e h a v i o r , but the d i f f i c u l t i e s b e g i n when one a t t e m p t s to apply a p a r t i c u l a r s o l u t i o n m o d e l to a wide r a n g e of alloy s y s t e m s w h e r e it is r e a l l y too much to expect that a s s u m p t i o n s made in the t h e o r e t i c a l m o d e l a r e valid in a l l c a s e s . What is needed is a specific solution m o d e l for each a l l o y s y s
Data Loading...