Effect of heat treatment on anodization and electrochemical behavior of AZ91D magnesium alloy
- PDF / 3,266,579 Bytes
- 9 Pages / 612 x 792 pts (letter) Page_size
- 97 Downloads / 354 Views
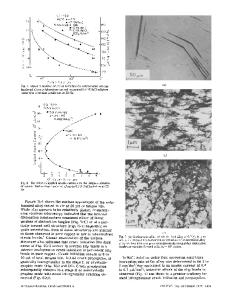
The anodic films formed on AZ91D magnesium alloy after heat treatment were analyzed and their electrochemical properties were investigated. The results showed that the cooling rate had a significant influence on the microstructure evolution of the AZ91D magnesium alloy after solution heat treatment at 440 °C for 20 h in N2 atmosphere. A single-phase microstructure was observed when the alloy was quenched in water after solution heat treatment. However, a duplex structure consisting of both ␣ and  phases was found if the solution-annealed alloy was cooled in air. The differences in microstructure of the heat treated AZ91D magnesium alloy gave rise to a significant change in the property of the anodic film formed in 3 M KOH + 0.21 M Na3PO4 + 0.6 M KF + 0.15 M Al(NO3)3 electrolyte. During the early stage of anodization, for the as-cast alloy, inhomogeneous anodic films were formed exhibiting relative rough surface appearances. A rather smooth anodic film was formed for the solution-annealed AZ91D magnesium alloy either followed by air cooling or water-quenched. The surface and cross section appearance was almost the same regardless of the prior heat treatment after anodizing for 20 min. The corrosion resistances of the various anodized AZ91D magnesium alloy were evaluated and compared by employing electrochemical impedance spectroscopy (EIS). The results demonstrate that the anodic film formed on the water-quenched AZ91D magnesium alloy had a slightly higher polarization resistance than that formed on the as-cast alloy. The highest polarization resistance of anodic film was found for that formed on annealed and air-cooled alloy. The presence of Al-rich  phase on the surface gave rise to the formation of a more protective anodic film which consisted of a great amount of Al2O3.
I. INTRODUCTION
The dependence of corrosion resistance of magnesium alloys on microstructure has been of interest and studied extensively. For AZ91D magnesium alloy which exhibits duplex microstructure, Linder et al.1 reported that the intermetallic Mg17Al12 precipitates ( phase) is in general more resistant to corrosion than the ␣ matrix. The high Al content in the intermetallic Mg17Al12 is the main factor giving rise to the high corrosion resistance of  phase. However, depending on its volume fraction and distribution,  phase can act as a corrosion barrier for the matrix or a galvanic cathode for the surrounding ␣ matrix.2,3 The casting method is also important in determining the corrosion performance of AZ91D magnesium
a)
Address all correspondence to this author. e-mail: [email protected] DOI: 10.1557/JMR.2005.0349 J. Mater. Res., Vol. 20, No. 10, Oct 2005
http://journals.cambridge.org
Downloaded: 14 Mar 2015
alloy. Ambat et al.4 indicated that die-cast AZ91D had higher corrosion resistance and better passivation than ingot-cast AZ91D because the former had fine grain structure and  phase. Song and co-workers have reported that the skin of die-cast AZ91D exhibited better corrosion resistance than its interior.3 Clearly, the
Data Loading...