Synthesis and Characterization of Photocatalytic TiO 2 -ZnFe 2 O 4 Nanoparticles
- PDF / 247,905 Bytes
- 6 Pages / 612 x 792 pts (letter) Page_size
- 91 Downloads / 520 Views
R5.9.1
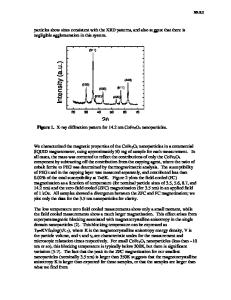
Synthesis and Characterization of Photocatalytic TiO2-ZnFe2O4 Nanoparticles Sesha S. Srinivasan, Jeremy Wade and Elias K. Stefanakos Clean Energy Research Center, College of Engineering, University of South Florida, Tampa, FL, 33620, U.S.A. ABSTRACT The wide bandgap semiconductor TiO2 has become the dominant UV-activated photocatalyst in the field of air and water detoxification because of its high stability, low cost, high oxidation potential and chemically favorable properties. The demand for visible-light activated photocatalytic systems is increasing rapidly; however, currently, the efficiency and availability of photocatalysts that can be activated effectively by the solar spectrum and particularly indoor lighting is severely limited. In this paper, a new coprecipitation/hydrolysis synthesis route is used to create a TiO2-ZnFe2O4 nanocomposite that is directed towards extending the photoresponse of TiO2 from UV to visible wavelengths (>400nm). The effect of TiO2’s accelerated anatase-rutile phase transformation due to the presence of the coupled ZnFe2O4 narrow bandgap semiconductor is evaluated. The transformation’s dependence on pH, calcination temperature, particle size, and ZnFe2O4 concentration has been analyzed using XRD, SEM, and UV-Visible spectrometry. The requirements for retaining the highly photoactive anatase phase present in a ZnFe2O4 nanocomposite are outlined. The visiblelight activated photocatalytic activity of the TiO2-ZnFe2O4 nanocomposites have been compared to an Aldrich TiO2 reference catalyst, using a solar-simulated photoreactor for the degradation of phenol. INTRODUCTION Environmental pollution on a global scale is proposed to be the greatest problem that chemical scientists will face in the 21st century, and an increasing number of these scientists are looking to new photocatalytic systems for the solution. The vast majorities of current photocatalytic system use pure or modified TiO2 with a metastable anatase crystal structure (3.2eV bandgap), although two key shortcomings exist. The first shortcoming is low photocatalytic efficiencies that plague current photocatalysts due to undesired electron-hole pair (EHP) recombination, and the second is that TiO2 utilizes only 3-5% of the solar spectrum and virtually none of the light commonly used for indoor illumination. Both of these spectral regions have applications needing active photocatalysts [1]. The push towards extending the photoresponse of TiO2 to visible wavelengths is increasing exponentially every year, for both solar (λ>UV-A,320nm) and visible light applications (λ>400nm). The most successful techniques used thus far for the development of modified TiO2 for visible-light photocatalysts are ion implantation methods using Cr or V ions [2], various synthesis techniques [3], and substitutional doping of non-metals such as N (TiO2-xNx) [4,5].
R5.9.2
Recent efforts have also been sought to extend the photoresponse of TiO2 through charge transfer interactions with narrow bandgap metal oxides such as the n-type ZnFe2O4 with
Data Loading...