Influence of the annealing atmosphere on solution based zinc oxide thin film transistors
- PDF / 217,622 Bytes
- 7 Pages / 432 x 648 pts Page_size
- 116 Downloads / 404 Views
Influence of the annealing atmosphere on solution based zinc oxide thin film transistors C. Busch1, R. Theissmann1, S. Bubel1, G. Schierning1and R. Schmechel1 Faculty of Engineering and Center for NanoIntegration Duisburg-Essen (CeNIDE), University of Duisburg-Essen, Bismarckstr. 81, D-47057 Duisburg, Germany.
1
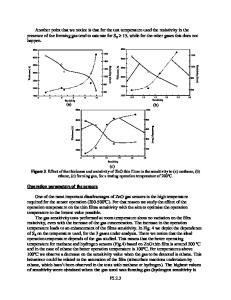
ABSTRACT Zinc oxide layers with a thickness of less than 10 nanometers have been synthesized from an aqueous solution for the application as active layer in thin film transistors. They have been conditioned by applying different oxidizing and reducing atmospheres during an annealing process at a temperature of 125°C. It is shown that the charge carrier mobility and threshold voltage is strongly influenced by the annealing atmosphere. Samples annealed in 10% forming gas (H2 in N2 - reducing atmosphere) show the highest field-effect-mobility of 0.6 cm2V-1s-1, but no saturation of the drain current, due to a high free carrier concentration. Samples treated under oxygen (strongest oxidizing atmosphere) show significantly lower mobilities. Subsequently, the samples have been exposed to synthetic air, with varying exposure times. Samples which have been annealed under hydrogen atmospheres show a pronounced decay of the drain current if exposed to synthetic air, whereas all samples conditioned under hydrogen-free atmospheres are significantly more stable under synthetic air. This enhanced sensitivity against oxygen after hydrogen treatment is attributed to residual hydrogen content in the sample that supports the formation of OH-groups which act as electron acceptors. INTRODUCTION Zinc oxide (ZnO) is a wide band gap semiconductor with acceptable electron mobilities and therefore a promising candidate for transparent, air-stable electronic devices like thin film transistors (TFTs), e.g. for application in active matrix flat panel displays or in radio frequency identification (RFID) tags [1, 2]. In order to deposit large areas with the desired material, printing or spin-coating are the most applicable methods for low-cost production of such semiconducting thin films with a thickness of only a few nanometers. Commonly, dispersions of nanoparticles or nanorods are used as the liquid precursors [3, 4]. A disadvantage of this approach is the use of stabilizers in the dispersion needed to avoid the agglomeration of the particles. However, these stabilizers hinder the conductivity of the layers, because of their electrically insulating properties [5]. In order to obviate this disadvantage, liquid precursors or solutions have been developed which are transformed into the desired ZnO-layers by a temperature treatment [6, 7]. While usually temperatures well above 200°C are required in order to decompose the precursors, Meyers et al [6] presented an ammonia complex Zn(OH)x(NH3)y(2-x)+ which decomposes well below 200°C. This offers the possibility to use flexible, transparent plastic substrates. In order to obtain this complex, Meyers et al. dissolved zinc nitrate in a caustic soda solution to obtain zinc hydroxide. Subsequen
Data Loading...