Twenty-four-month results of intravitreal aflibercept for macular edema due to branch retinal vein occlusion
- PDF / 752,577 Bytes
- 6 Pages / 595.276 x 790.866 pts Page_size
- 28 Downloads / 310 Views
CLINICAL INVESTIGATION
Twenty‑four‑month results of intravitreal aflibercept for macular edema due to branch retinal vein occlusion Yoshihito Sakanishi1 · Koichi Yasuda1 · Syu Morita1 · Keitaro Mashimo1 · Kazunori Tamaki1 · Toshiro Sakuma1 · Nobuyuki Ebihara1 Received: 3 June 2020 / Accepted: 25 September 2020 © Japanese Ophthalmological Society 2020
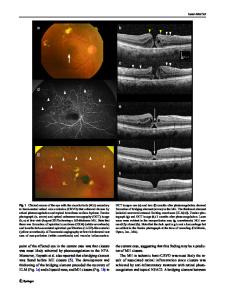
Abstract Purpose To investigate 24-month results of intravitreal aflibercept (IVA) for macular edema due to branch retinal vein occlusion (BRVO-ME). Study design Retrospective study. Methods Subjects were treatment-naïve BRVO-ME patients at the Ophthalmology Department of Juntendo University Urayasu Hospital from November 2015 to March 2017 who received IVA treatment for 24 months. After the first injection, reinjection was performed as needed when ME had recurred or was prolonged beyond 300 μm. Data included changes in best corrected visual acuity and central foveal thickness, total number of injections, and the case background factors that required reinjection after 12 months. ME remission was defined as patients without additional injections for 6 months. Results Forty eyes of 40 patients (64.5 ± 11.5 years), 21 men and 19 women, were included. Average best corrected visual acuity and central foveal thickness at baseline were logMAR 0.42 ± 0.21, 601.4 ± 181.3 μm, logMAR 0.08 ± 0.25, 214.6 ± 62.7 μm at 12 months, logMAR 0.02 ± 0.16, 216.6 ± 97.8 μm at 24 months, all significantly improved from baseline. The average number of injections was 2.2 ± 1.0 times in the first year and 0.4 ± 0.8 times in the second year. The rate of ME remission was 60.0% at 12 months and 87.5% at 24 months. Period between onset and injections was significantly associated with reinjection after 12 months (p =.030). Conclusions IVA was effective over 24 months for ME due to BRVO in many cases. Early injection treatment may reduce the need for later injections. Keywords BRVO · Macular edema · IVA
Introduction Retinal vein occlusion is the second most common retinal vascular disease after diabetes mellitus retinopathy [1, 2]. Branch retinal vein occlusion (BRVO) and central retinal vein occlusion are classified according to the site of vascular occlusion, and both induce vision impairment due to macular edema (ME). Based on the results of a large-scale study, retinal photocoagulation has been used as treatment for BRVO, but visual acuity improvements were limited [3]. Corresponding Author: Yoshihito Sakanishi * Yoshihito Sakanishi [email protected] 1
Department of Ophthalmology, Juntendo University Urayasu Hospital, 2‑1‑1 Tomioka, Urayasu, Japan
AS vascular endothelial growth factor (VEGF) was deemed responsible for ME, treatment with anti-VEGF drugs was introduced. First, bevacizumab, originally approved as an anticancer drug for colorectal cancer, was used in intravitreal injections off-label [4]. Bevacizumab is a humanized monoclonal antibody that inhibits angiogenesis by inhibiting VEGF-A and has shown good results treating ME [5]. Subsequently, ranibizumab, a humanized
Data Loading...