NASICON-like phases in ScPO 4 -Na 3 PO 4 -Li 3 PO 4 quasiternary system
- PDF / 91,306 Bytes
- 6 Pages / 612 x 792 pts (letter) Page_size
- 90 Downloads / 469 Views
0936-L01-06
NASICON-like phases in ScPO4-Na3PO4-Li3PO4 quasiternary system Mariya Zhuravleva1, Ruslan Zakalyukin1, Valeriy Fomichev1, Andrey Novoselov2, Akira Yoshikawa2, Tsuguo Fukuda2, and Galina Zimina1 1 Lomonosov Moscow State Academy of Fine Chemical Technology, Vernadskogo Av. 86, Moscow, 119571, Russian Federation 2 Division of Advanced Crystal Materials, Institute of Multidisciplinary Research for Advanced Materials, Tohoku University, Katahira 2-1-1, Aoba-ku, Sendai, 980-8577, Japan
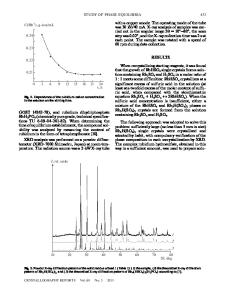
ABSTRACT Phase formation in ternary system of complex Sc, Na and Li phosphates was studied at 950°C and synthesis of new phases of definite composition was carried out. Obtained samples were investigated with X-ray powder diffraction, infrared and impedance spectroscopy. Compositions of fields of homogeneity with the NASICON-like structure were discovered. Temperature dependency of ionic conductivity was measured.
INTRODUCTION There is a challenge of finding new solid-state electrolytes having high values of ionic conductivity at low (room) working temperatures. Solid-state electrolytes are materials that have high ion mobility due to their unique crystal structure where ions can move easily through interstitials. High values of ionic conductivity are achieved by migration of alkaline cations. Such compounds as β-alumina, α-AgI and their derivatives with high values of ionic conductivity are well known. One of the promising materials for solid electrolytes, phosphosilicate of sodium and zirconium Na1+xZr2SixP3−xO12, was discovered by Hong and co-workers [1, 2]. The best electrical properties are found for Na3Zr2Si2PO12 called NASICON (NAtrium Super Ionic CONductor), that exhibits a higher conductivity than β-alumina above 300°C (σ = 0.25 Ω−1·cm−1). These materials lend themselves to a wide range of applications, for example in solid-state batteries, fuel cells and gas sensors [3]. A number of NASICON-like compounds have been intensively studied [4-6]. Later, it was found that the binary system of ScPO4-Na3PO4 also includes NASICON-like compounds: Na3Sc2(PO4)3 and Na6Sc3(PO4)5 [7]. They possess relatively high values of ionic conductivity which can be understood by their three-dimensional structure [8]. Its framework consists of (PO4)-tetrahedra sharing corners with (ScO6)-octahedra with interstitial space occupied by Na+-cations. Significant ionic conductivity of Na3Sc2(PO4)3 (10-1 Ω−1·cm−1) is achieved only at high temperature of 300°C that limits its practical application. Properties of NASICON-like compounds are characterized by the statistical nature of the filling A- and B-type cavities by small Na+-cations and their mobility in the system of channels [9]. These compounds have complex polymorphism, for example Na3Sc2(PO4)3 has 3 polymorphous modifications: α → β (~ 50°C), β → γ (~ 168°C) [10].
High values of ionic conductivity can be also achieved by Li cations. Li3Sc2(PO4)3 has 3-dimensional structure with Li+-cation conductivity around 10-3-10-2 Ω−1·cm−1 at 300°C [11]. Li cations fill their crystallographic positions
Data Loading...